- Blog
- About
- Contact
- Mcafee mac os 10-11
- Run appium app on android device
- Watch goodnight mommy part 1
- Stellar phoenix mac data recovery torrent
- Apple mac pro reviews
- Sketchup 2018 crack mac reddit
- Can you upgrade mac mini graphics card
- Ready to die biggie smalls izp
- Download old version of manycam for mac
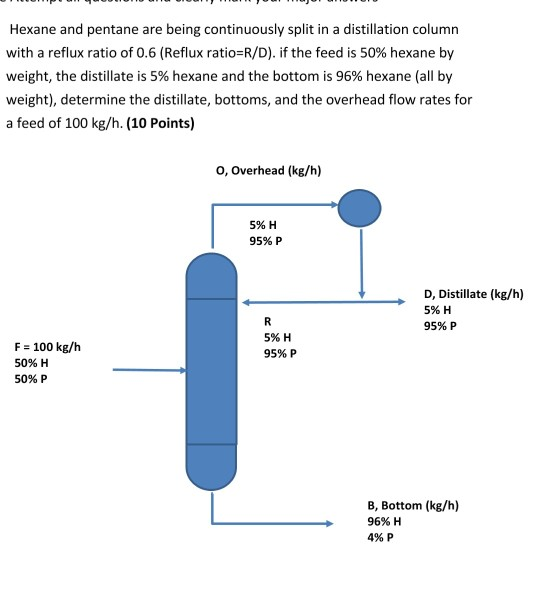
- Relux ratio
Packed distillation columns use specialized packing material with small channels to increase liquid-vapor contact in smaller quarters. There must also be sufficient interfacial area on any given stage. In order to make the partial vaporization in each tray into a practical separation process, the liquid and vapor must flow countercurrently through several stages. In a distillation column, density differences cause vapor to flow upward to the stage above, and gravity causes the liquid to flow downward to the tray below. 2Īll distillation processes depend upon composition differences between vapor and liquid phases in contact that are exchanging mass. Any Separations or Unit Operations textbook covers the fundamentals of distillation including basic concepts such as reflux ratio, Murphree efficiencies and the McCabe-Thiele method and diagram.

Additionally, data reconciliation of the mass balances can be performed to determine if gross measurement errors exist. These two methods can also be used to determine the overall tray efficiency. The results can also be simulated using an equilibrium process simulator, if available. From the sample analyses, the tray efficiencies can be determined for all three components on all six trays assuming constant molar overflow (McCabe-Thiele method). A typical protocol is to investigate the effects of reflux ratio over a wide range. Once steady state is achieved (takes at least 90 min), liquid samples will be taken from the reflux drum, reboiler and on each tray and analyzed in a gas chromatograph. After a steady reflux drum level is attained, a switch to finite reflux mode is made by adjusting the bottoms, distillate and reflux flow rate controllers as necessary to maintain steady levels in the reflux drum and the reboiler, and to maintain a target reflux ratio R D = L/D. In this demonstration, the Tray Distillation Unit (TDU) is started in total reflux mode. A P&ID schematic of the sieve tray (each tray contains holes in a support plate) distillation system can be found in Appendix A. Sieve trays provide maximum interfacial area between the liquid and vapor.

1, 2 The goal of this experiment is to distill a mixture of alcohols (methanol, isopropanol) and water in a sieve tray column and examine how closely simple theories of distillation based on equilibrium assumptions are followed. Tray and packed columns are both commonly used for distillation, absorption, and stripping separation operations. Benton, Department of Chemical Engineering, Louisiana State University, Baton Rouge, LA
